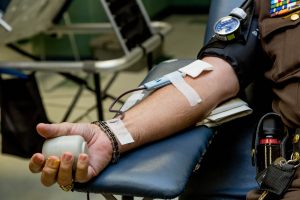
Recently, the President announced that the FDA had approved the emergency use of convalescent plasma treatment for COVID-19 patients. There have been indications that this could be an effective treatment, but experts caution that we do not have sufficient data to confirm this.
Treatment and Trials Of Plasma Treatment
The convalescent plasma treatment involves the infusion of the part of the blood that has the most virus antibodies from recovered patients of COVID-19 into patients who are currently battling the illness. The treatment has been tested on around 70,000 COVID patients and has shown positive results and a drop in mortality, but a proper study involving a placebo to see the difference has not been conducted. There is also a concern that since the EUA allows for more patients to receive the treatment, fewer patients will sign up for a clinical trial where they may receive a placebo. This has the potential to make it difficult to confirm the effectiveness of the treatment.
Politicization and Approval Of Plasma Treatment
During the White House announcement about this emergency use authorization, the president took to throwing stones at the FDA for delaying the approval for suspected political reasons. Despite these assertions, the head of the FDA says that the doctors at the FDA make all their decisions based on data that they have available. There have been rumors that the emergency use authorization was granted due to pressure from the White House to speed up the process.
Moving Too Fast?
Medical options being pushed out too quickly can often lead to negative consequences, as negative side effects and potential ineffectiveness could arise. However, with this treatment, it is confirmed to be safe but may not be as effective as we hope. This determination of safety is what has allowed it to be approved without complete trials.
Insurance Questions?
Empower wants to help you get the best insurance for your needs and the most for your money. We want to help you stay on top of your health.
CALL US at (844) 410-1320
Get affordable health insurance quotes by clicking here.
See our other websites:
EmpowerLifeInsurance.com
EmpowerMedicareSupplement.com
EmpowerMedicareAdvantage.com

