In late August, President Trump announced that the Food and Drug Administration (FDA) had completed the authorization process for convalescent plasma treatment to aid victims of the coronavirus pandemic. The COVID-19 plasma treatment was previously available to many Americans, yet the FDA’s authorization will make the treatment more accessible to a wider array of patients.
Why now?
“Scientists have concluded that COVID-19 convalescent plasma is safe and shows promising efficacy, thereby meeting the criteria for an emergency use authorization,” stated FDA Commissioner Stephen Hahn at the August 23 briefing. With no coronavirus cure yet, the FDA approved the treatment as an emergency measure.
How does the COVID-19 plasma treatment work?
Convalescent plasma therapy consists of separating plasma from blood donations of COVID-19 survivors and using it to treat other patients. Because the survivors’ blood contains powerful antibodies that help their bodies fight the virus, the patients’ bodies are better equipped to fight COVID.
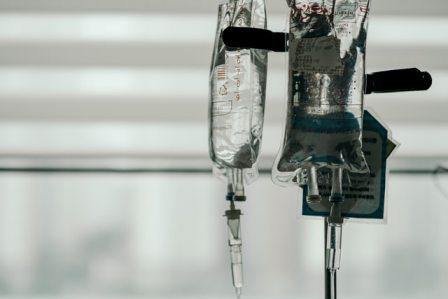
The plasma is yellow and is frozen until use. Patients receiving convalescent plasma therapy are closely monitored both during and after the procedure to ensure that no side effects appear. The plasma is administered through an intravenous, or IV, tube. The fluid drips from the sterile sack it is stored in and into the patient’s bloodstream. The entire procedure takes around two hours to complete.
What studies have been conducted?
Some researchers are concerned that the treatment, since it was already so readily available, was too difficult to accurately analyze in randomized case studies. Studies like the Michigan-based Clinical Trial of COVID-19 Convalescent Plasma of Outpatients, lightheartedly referred to as C3PO, however, have provided valuable data that may have led the FDA to this decision.
As a randomized controlled trial where half the enrolled patients receive plasma and half receive a saline solution, C3PO proved that the COVID-19 plasma treatment helps patients, to some extent. As of September 1, the Biomedical Advanced Research and Development Authority’s (BARDA) US COVID Plasma effort has treated 87,000 patients nationwide in 2,700 hospitals.
As of late August, the COVID-19 plasma treatment showed a 35 percent improvement in the survival rate of coronavirus patients. Thousands of patients have received antibodies via plasma treatment throughout the coronavirus pandemic. No data suggest that plasma treatments are an effective COVID-19 treatment, but small clinical trials and data from a national access program suggest that the procedure may help lessen the symptoms or shorten the duration of COVID-19 cases.
Health Insurance Questions?
We hope this information on the new COVID-19 plasma treatment is helpful.
Empower Brokerage wants to help you understand the insurance coverage you need and how to save money getting it. Stay on top of your health and give us a call at (844) 410-1320
Get affordable health insurance quotes by clicking here.
See our other websites:

